
Director of Research
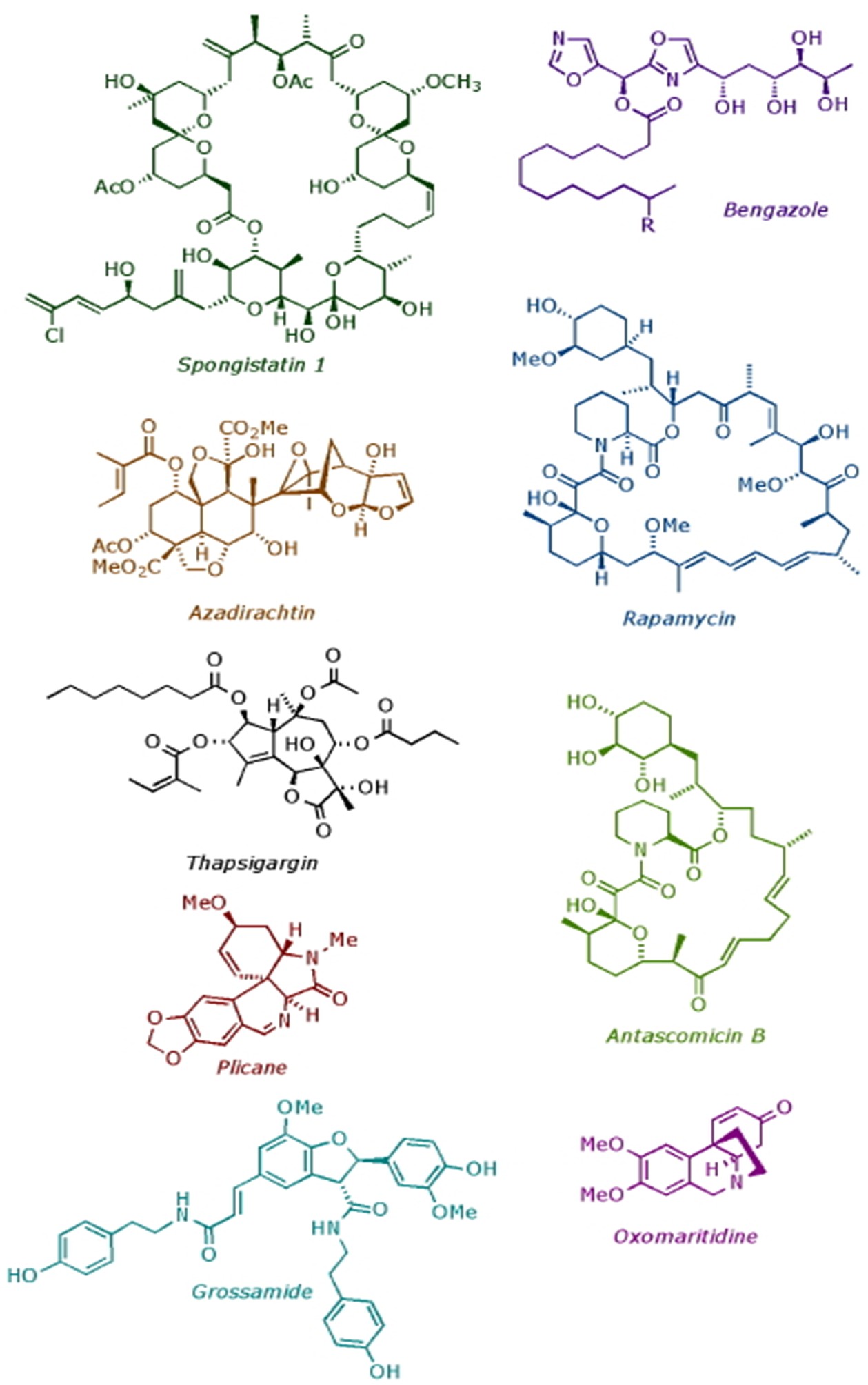
In the Ley Group, we specialise in developing new synthesis methods and applying them to the construction of biologically important molecules. Over the years we have completed the total synthesis of many natural products, including: spongistatin 1 (anti-mitotic agent); rapamycin (immunosuppressant); thapsigargin (SERCA pumps inhibitor); azadirachtin (insect antifeedant); and bengazole A (fungicide). In addition to our research on natural product synthesis, we also pioneered flow chemistry and machine assisted synthesis.
For more detailed research information and our publication list, please see our legacy group website.
Completed Natural Products
Publications
CHEMISTRY OF INSECT ANTIFEEDANTS FROM AZADIRACHTA-INDICA .17. SYNTHESIS OF MODEL COMPOUNDS OF AZADIRACHTIN - UNUSUAL EFFECT OF REMOTE SUBSTITUENTS ON THE COURSE OF THE OXIDATIVE RING CONTRACTION REACTION
Tetrahedron
(1994)
50
11553
SELECTIVE ACYLATION AND ALKYLATION REACTIONS OF DIOLS USING DIBUTYLTIN DIMETHOXIDE (PG 913, 1993)
SYNLETT
(1994)
764
CHEMISTRY OF INSECT ANTIFEEDANTS FROM AZADIRACHTA-INDICA .16. SYNTHESIS OF SEVERAL DERIVATIVES OF AZADIRACHTIN CONTAINING FLUORESCENT OR IMMUNOGENIC REPORTER GROUPS
Tetrahedron
(1994)
50
8871
Sexual development of malaria parasites is inhibited in vitro by the Neem extract Azadirachtin, and its semi-synthetic analogues
FEMS Microbiology Letters
(1994)
120
267
(doi: 10.1016/0378-1097(94)90482-0)
Model studies towards the insect antifeedant Jodrellin A using an organoselenium mediated cyclization reaction
Tetrahedron Letters
(1994)
35
4861
Dispiroketals in synthesis (part 10): Further reactions of dispoke protected lactate and glycolate enolates
Tetrahedron
(1994)
50
7157
Studies towards the total synthesis of rapamycin: Preparation of the cyclohexyl C33C42 fragment and further coupling to afford the C22C42 carbon unit
Tetrahedron Letters
(1994)
35
2091
Studies towards the total synthesis of rapamycin: Preparation of the C10C17 carbon unit
Tetrahedron Letters
(1994)
35
2095
Studies towards the total synthesis of rapamycin: A convergent and stereoselective synthesis of the C22C32 carbon framework
Tetrahedron Letters
(1994)
35
2087
Dispiroketals in synthesis (part 7): Protection of D-glucopyranose substrates
Tetrahedron Letters
(1994)
35
773
- ‹ previous
- Page 79

