
Director of Research
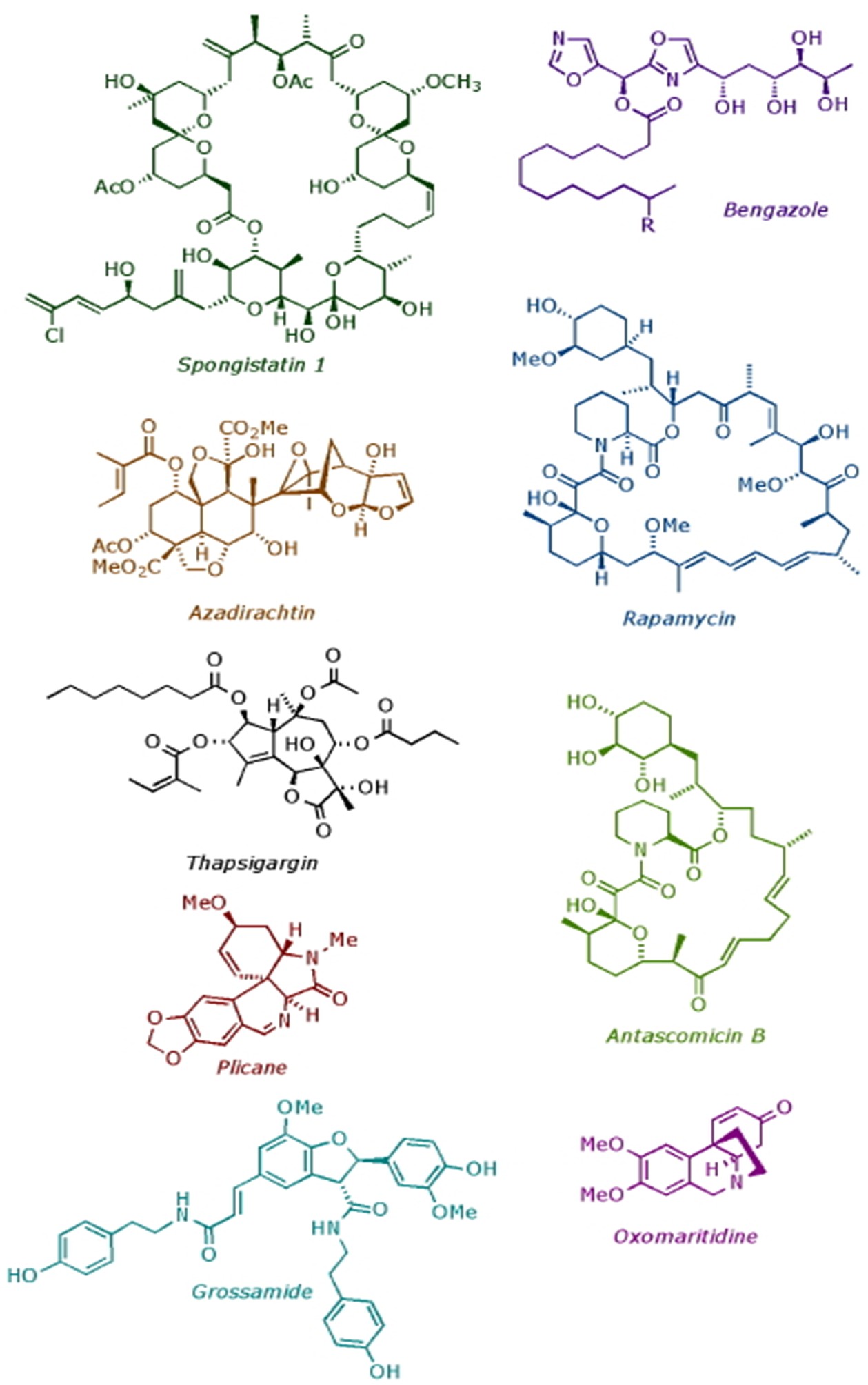
In the Ley Group, we specialise in developing new synthesis methods and applying them to the construction of biologically important molecules. Over the years we have completed the total synthesis of many natural products, including: spongistatin 1 (anti-mitotic agent); rapamycin (immunosuppressant); thapsigargin (SERCA pumps inhibitor); azadirachtin (insect antifeedant); and bengazole A (fungicide). In addition to our research on natural product synthesis, we also pioneered flow chemistry and machine assisted synthesis.
For more detailed research information and our publication list, please see our legacy group website.
Completed Natural Products
Publications
Biosynthesis of tetronasin: Part 4, preparation of deuterium labelled C19-C26, C17-C26, C11-C26 and C3-C26 polyketide fragments as putative biosynthetic precursors of the ionophore antibiotic tetronasin (ICI 139603)
Tetrahedron
(1995)
51
5417
(doi: 10.1016/0040-4020(95)00201-i)
CHEMISTRY OF INSECT ANTIFEEDANTS FROM AZADIRACHTA-INDICA .18. DEMETHYLATION AND METHYLATION OF THE C-8 POSITION OF THE DECALIN PORTION OF AZADIRACHTIN
Tetrahedron
(1995)
51
2077
(doi: 10.1016/0040-4020(94)01071-7)
Behavioural responses of locusts and Spodoptera littoralis to azadirachtin and azadirachtin analogues containing fluorescent and immunogenic reporter groups
Journal of Insect Physiology
(1995)
41
555
(doi: 10.1016/0022-1910(95)00014-l)
Dispiroketals in synthesis (part 19)1: Dispiroketals as enantioselective and regioselective protective agents for symmetric cyclic and acyclic polyols.
Tetrahedron Asymmetry
(1995)
6
2403
(doi: 10.1016/0957-4166(95)00318-J)
Synthesis of β-dimorphecolic acid exploiting highly stereoselective reduction of a side-chain carbonyl group in a π-allyltricarbonyliron lactone complex
Chemical Communications
(1995)
1751
(doi: 10.1039/C39950001751)
“Connectivist” approach to organic structure determination Lsd-program assisted Nmr analysis of the insect antifeedant Azadirachtin
Tetrahedron
(1994)
50
12267
Dispiroketals in synthesis (Part 13): Functionalised dispiroketals as new chiral auxiliaries; highly stereoselective diels-alder reactions using a bifunctional, C2- symmetrical chiral auxiliary
Tetrahedron Letters
(1994)
35
7451
(doi: 10.1016/0040-4039(94)85339-8)
Dispiroketals in synthesis (Part 14): Functionalised dispiroketals as new chiral auxiliaries; highly stereoselective Michael additions to a bifunctional, C2- symmetrical chiral auxiliary
Tetrahedron Letters
(1994)
35
7455
(doi: 10.1016/0040-4039(94)85340-1)
Dispiroketals in synthesis (Part 12): Functionalised dispiroketals as new chiral auxiliaries; the synthesis of dihydroxylated dispiroketals in optically pure form
Tetrahedron Letters
(1994)
35
7447
(doi: 10.1016/0040-4039(94)85338-x)
Dispiroketals in synthesis (Part 11): Concomitant enantioselective and regioselective protection of 2,5- dibenzoyl-myo-inositol
Tetrahedron Letters
(1994)
35
7443
(doi: 10.1016/0040-4039(94)85337-1)
- ‹ previous
- Page 78

