
Director of Research
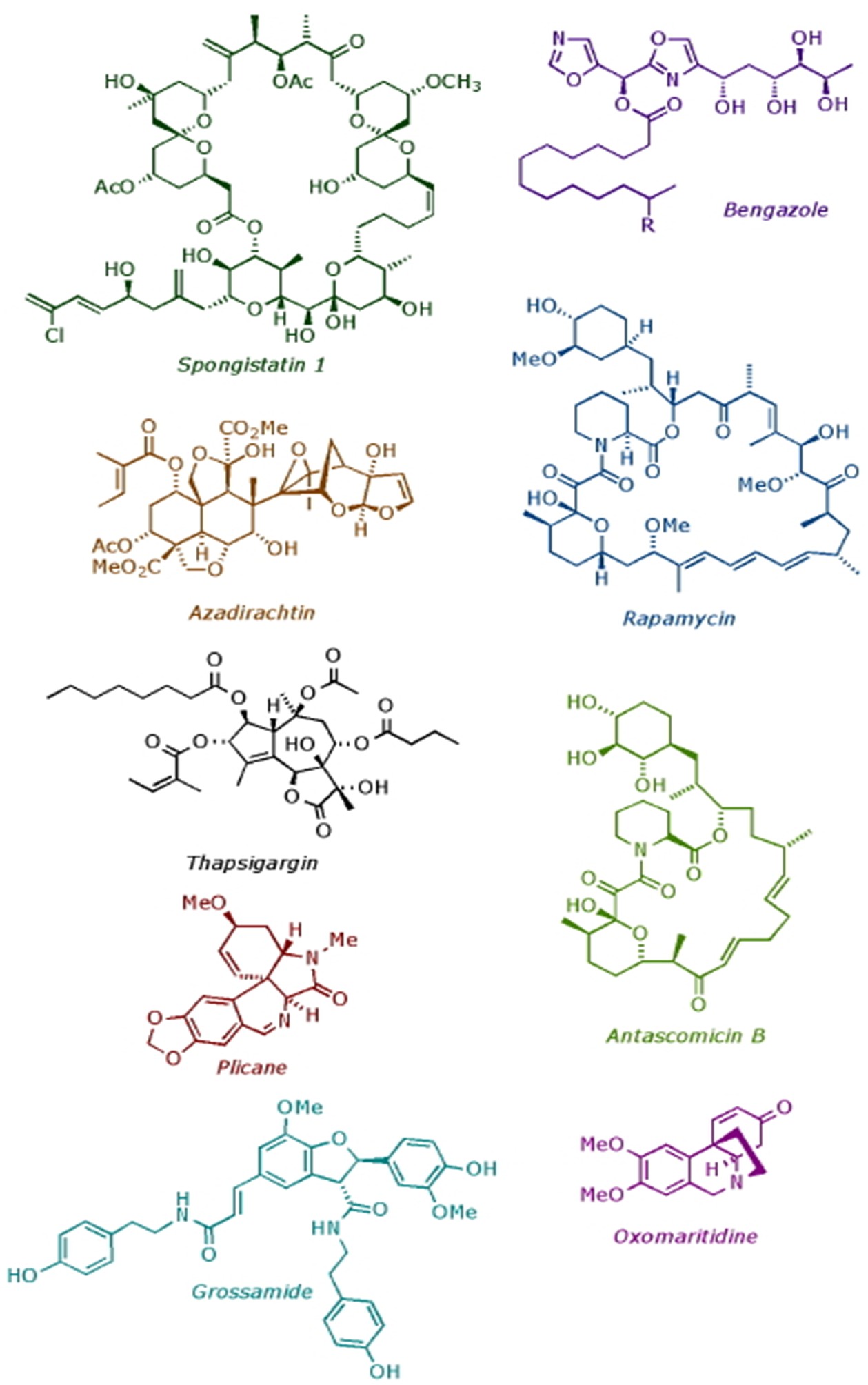
In the Ley Group, we specialise in developing new synthesis methods and applying them to the construction of biologically important molecules. Over the years we have completed the total synthesis of many natural products, including: spongistatin 1 (anti-mitotic agent); rapamycin (immunosuppressant); thapsigargin (SERCA pumps inhibitor); azadirachtin (insect antifeedant); and bengazole A (fungicide). In addition to our research on natural product synthesis, we also pioneered flow chemistry and machine assisted synthesis.
For more detailed research information and our publication list, please see our legacy group website.
Completed Natural Products
Publications
Chemo-enzymatic synthesis of fluorinated sugar nucleotide:: Useful mechanistic probes for glycosyltransferases
Bioorganic & Medicinal Chemistry
(2000)
8
1937
Protecting groups: Effects on reactivity, glycosylation stereoselectivity, and coupling efficiency
(2000)
1-4
427
(doi: 10.1002/9783527618255.ch17)
Synthesis of High-Mannose Type Neoglycolipids: Active Targeting of Liposomes to Macrophages in Gene Therapy
Chemistry (Weinheim an der Bergstrasse, Germany)
(2000)
6
1416
Asymmetric molybdenum-catalyzed allylic substitution.
ABSTRACTS OF PAPERS OF THE AMERICAN CHEMICAL SOCIETY
(2000)
219
U132
Pinene-derived bipyridine ligands (PINDY) in asymmetric catalysis.
ABSTR PAP AM CHEM S
(2000)
219
U132
Polymer supported reagents in synthesis: preparation of bicyclo[2.2.2] octane derivates via Tandem Michael addition reactions and subsequent combinatorial decoration.
Journal of combinatorial chemistry
(2000)
2
104
(doi: 10.1021/cc9900697)
Rapid assembly of oligosaccharides: 1,2-diacetal-mediated reactivity tuning in the coupling of glycosyl fluorides
Tetrahedron: Asymmetry
(2000)
11
173
A short and efficient stereoselective synthesis of the polyhydroxylated macrolactone (+)-aspicillin.
Organic letters
(2000)
2
123
(doi: 10.1021/ol991214s)
Deracemization of Baylis-Hillman adducts
Chemtracts
(2000)
13
596
A short and efficient stereoselective synthesis of the potent 5-lipoxygenase inhibitor CMI-977
Synthetic Communications
(2000)
30
1955
(doi: 10.1080/00397910008087245)
- ‹ previous
- Page 65

