
Director of Research
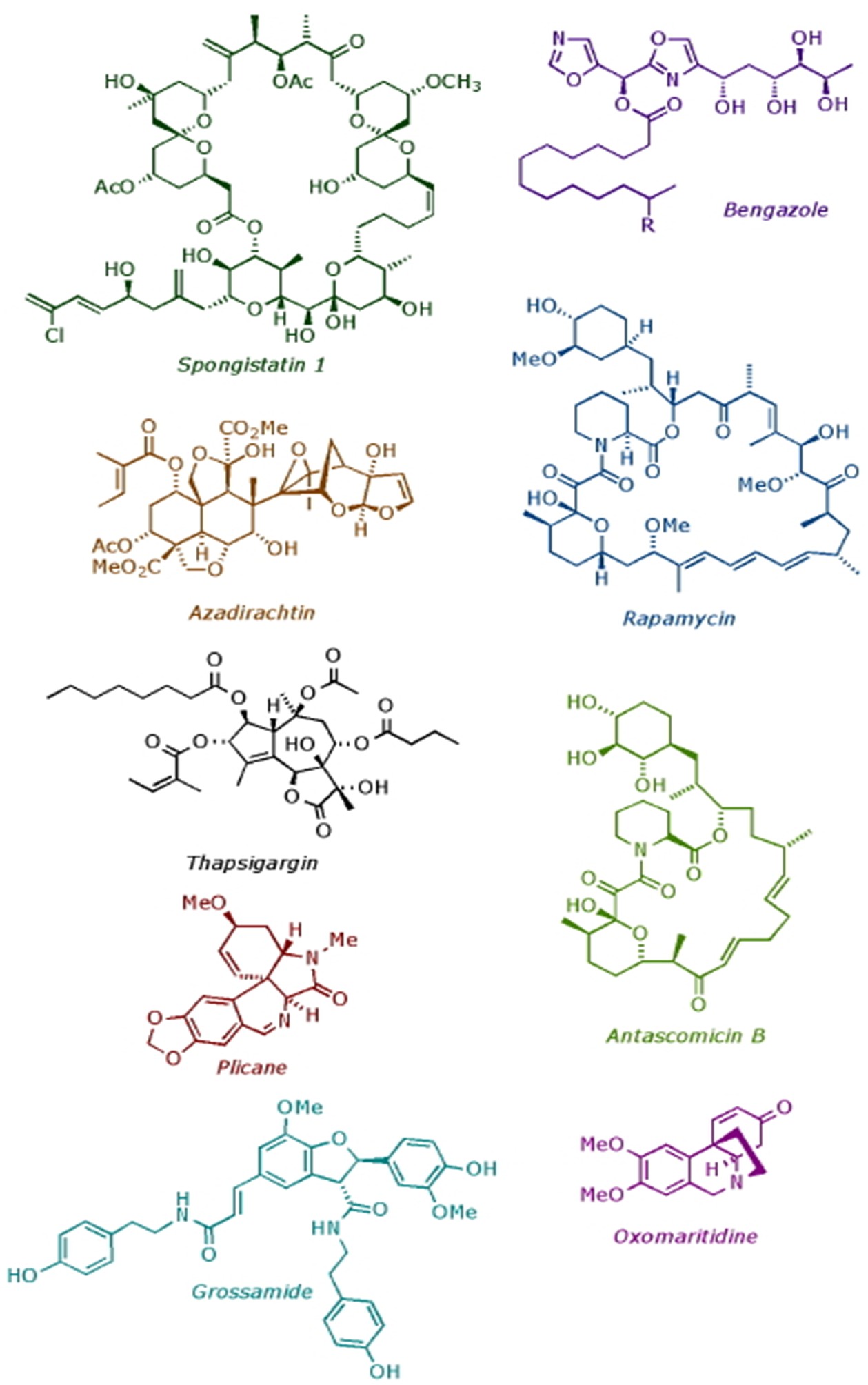
In the Ley Group, we specialise in developing new synthesis methods and applying them to the construction of biologically important molecules. Over the years we have completed the total synthesis of many natural products, including: spongistatin 1 (anti-mitotic agent); rapamycin (immunosuppressant); thapsigargin (SERCA pumps inhibitor); azadirachtin (insect antifeedant); and bengazole A (fungicide). In addition to our research on natural product synthesis, we also pioneered flow chemistry and machine assisted synthesis.
For more detailed research information and our publication list, please see our legacy group website.
Completed Natural Products
Publications
Preparation of butane-1,2-diacetal-protected l-glyceraldehyde from D-mannitol
Synthesis
(2003)
2004
147
(doi: 10.1055/s-2003-42489)
A Practical and Efficient Synthesis of the C-16−C-28 Spiroketal Fragment (CD) of the Spongistatins
Organic Letters
(2003)
5
4815
(doi: 10.1021/ol035848h)
Modern Synthetic Methods for Copper-Mediated C(aryl)-O, C(aryl)-N, and C(aryl)-S Bond Formation
Angew Chem Int Ed Engl
(2003)
42
5400
(doi: 10.1002/anie.200300594)
Palladium-containing perovskites: recoverable and reuseable catalysts for Suzuki couplingsElectronic supplementary information (ESI) available: experimental details. See http://www.rsc.org/suppdata/cc/b3/b308465e/
Chemical communications (Cambridge, England)
(2003)
9
2652
(doi: 10.1039/b308465e)
Recyclable polyurea-microencapsulated Pd(0) nanoparticles: An efficient catalyst for hydrogenolysis of epoxides
Org Lett
(2003)
5
4665
(doi: 10.1021/ol0358509)
A Convenient Route to Enantiomerically Pure 2-Substituted Methyl Glycerate Derivatives
Organic letters
(2003)
5
4553
(doi: 10.1021/ol035567+)
A sequential tetra-n-propylammonium perruthenate (TPAP)–Wittig oxidation olefination protocol
Tetrahedron Letters
(2003)
44
7779
(doi: 10.1016/j.tetlet.2003.08.081)
Total synthesis of the polyenoyltetramic acid polycephalin C
Tetrahedron
(2003)
59
6955
Synthesis, Chiroptical Properties, and Absolute Configuration of (+)-2,3-Dihydrotriquinacen-2-one. Effect of Rigid Triquinacene Geometry on the Inherently Dissymmetric Chromophore
Journal of the American Chemical Society
(2003)
97
7273
(doi: 10.1021/ja00858a600)
- ‹ previous
- Page 51

