
Director of Research
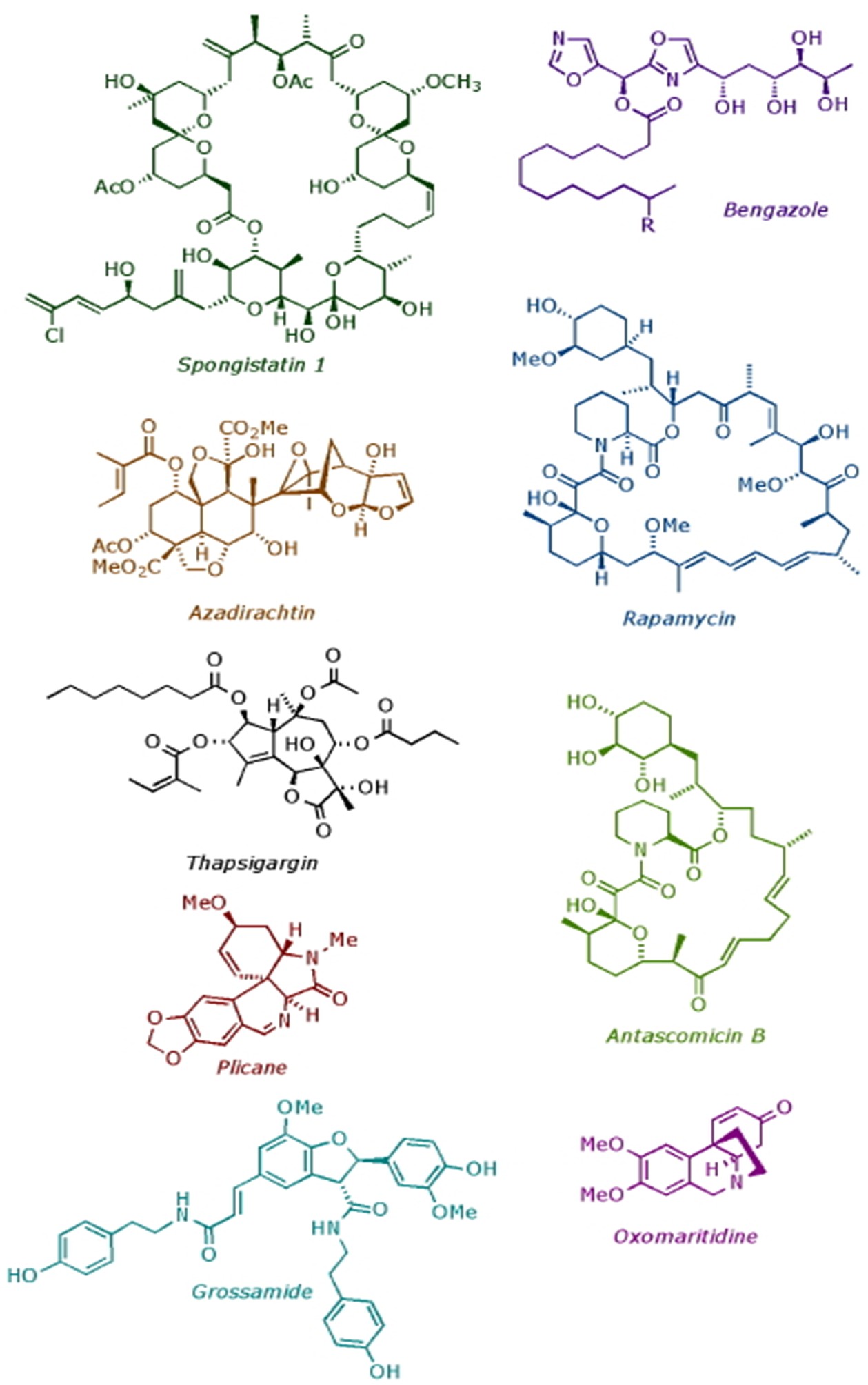
In the Ley Group, we specialise in developing new synthesis methods and applying them to the construction of biologically important molecules. Over the years we have completed the total synthesis of many natural products, including: spongistatin 1 (anti-mitotic agent); rapamycin (immunosuppressant); thapsigargin (SERCA pumps inhibitor); azadirachtin (insect antifeedant); and bengazole A (fungicide). In addition to our research on natural product synthesis, we also pioneered flow chemistry and machine assisted synthesis.
For more detailed research information and our publication list, please see our legacy group website.
Completed Natural Products
Publications
Highly cis- or trans-selective oxygen to carbon rearrangements of anomerically linked 6-substituted tetrahydropyranyl enol ethers
Journal of the Chemical Society Perkin Transactions 1
(1999)
2665
(doi: 10.1039/a904891j)
Modified mesoporous silicate MCM-41 materials: immobilised perruthenate—a new highly active heterogeneous oxidation catalyst for clean organic synthesis using molecular oxygen
Chemical Communications
(1999)
1907
(doi: 10.1039/a906354d)
Three-step synthesis of an array of substituted benzofurans using polymer-supported reagents
Journal of the Chemical Society Perkin Transactions 1
(1999)
2421
(doi: 10.1039/a904384e)
π-allyltricarbonyliron lactone complexes in a diastereoselective Mukaiyama aldol reaction.
ABSTR PAP AM CHEM S
(1999)
218
U51
Total synthesis of the plasmoidal pigment physarorubinic acid, a polyenoyl tetramic acid
Journal of the Chemical Society Perkin Transactions 1
(1999)
2231
(doi: 10.1039/a904921e)
Use of the Potassium Ion as a Template for the Selective Derivatization of the Antibiotic X-206
The Journal of Organic Chemistry
(1999)
64
6252
(doi: 10.1021/jo9903151)
Total synthesis of the cholesterol biosynthesis inhibitor 1233A via a (pi-allyl)tricarbonyliron lactone complex
J CHEM SOC PERK T 1
(1999)
1917
(doi: 10.1039/a903641e)
Synthesis of an array of potential matrix metalloproteinase inhibitors using a sequence of polymer-supported reagents.
Bioorganic & medicinal chemistry letters
(1999)
9
2049
Dispiroketals in synthesis.: Part 24.: Preparation and use of chiral 2,2′-bis(triisopropylsilyloxymethyl)bi(dihydropyran)s as new protecting and resolving agents for 1,2-diols
Journal of the Chemical Society Perkin Transactions 1
(1999)
1639
(doi: 10.1039/a900950g)
Dispiroketals in synthesis. Part 25.1 Further reactions of dispiroketal protected glycolate to afford optically active 1,2,3,4-tetraols
JOURNAL OF THE CHEMICAL SOCIETY-PERKIN TRANSACTIONS 1
(1999)
1647
(doi: 10.1039/a900951e)
- ‹ previous
- Page 67

